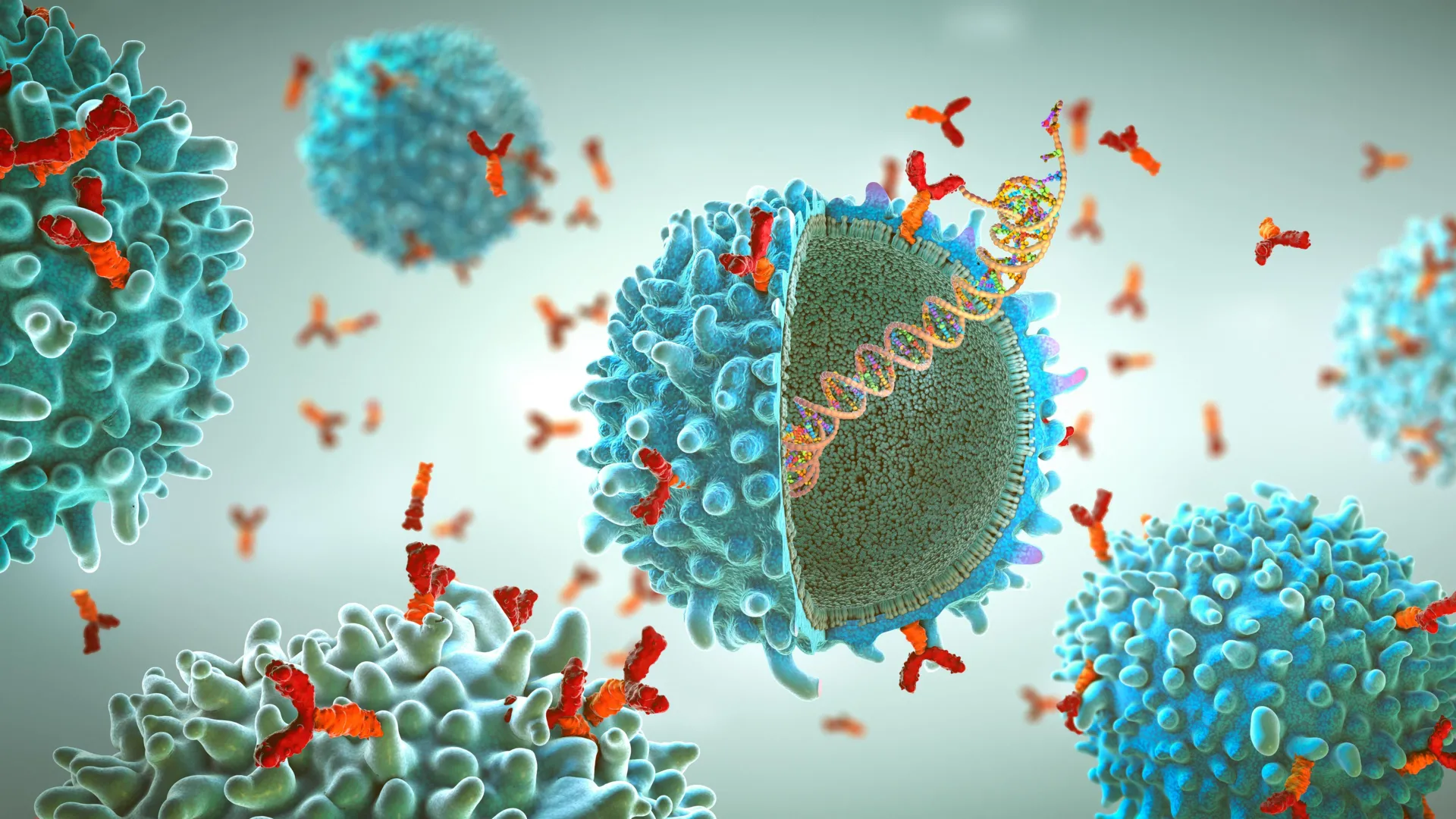
Viruses are highly effective at entering human cells, largely because of specialized proteins that cover their outer surfaces. These proteins are key targets in vaccine development. To study them, scientists typically create lab versions to see how the immune system might respond. However, these simplified versions often leave out important sections that normally sit within the virus’s outer membrane. As a result, they do not always behave the same way they would in a real infection, making it harder to understand how antibodies truly recognize and stop viruses.
Researchers at Scripps Research, working with IAVI and other collaborators, have now developed a new platform that allows these viral proteins to be studied in a much more natural form. Their method uses nanodisc technology, which places the proteins into tiny particles made of lipids. This setup mimics the virus’s outer membrane, helping preserve the proteins’ natural structure and behavior. The approach offers a clearer view of how antibodies interact with viruses and could help guide future vaccine design.
Nanodisc Technology Mimics Viral Membranes
The study, published in Nature Communications, tested the platform using proteins from HIV and Ebola. These viruses have long posed challenges for vaccine development because their surface proteins are especially difficult for the immune system to target. The researchers believe the same method could also be applied to other viruses with similar membrane-bound proteins, including influenza and SARS-CoV-2.
“For many years, we’ve had to rely on versions of viral proteins that are missing important pieces,” says co-senior author William Schief, a professor at Scripps Research and executive director of vaccine design at IAVI’s Neutralizing Antibody Center. “Our platform lets us study these proteins in a setting that better reflects their natural environment, which is critical if we want to understand how protective antibodies recognize a virus.”
In real viruses, surface proteins are embedded within a lipid membrane and arranged in specific shapes. In contrast, most laboratory studies remove the membrane-anchoring portion to make the proteins easier to handle. While this simplifies experiments, it can hide important details, especially for antibodies that target regions near the base of the protein close to the membrane.
To overcome this limitation, the team incorporated vaccine candidate proteins into nanodiscs. These small, stable lipid patches hold the proteins in place and closely resemble the virus’s outer layer. This setup allows scientists to study how antibodies interact with proteins in a more realistic context. The platform also supports standard vaccine research tools, including antibody binding tests, immune cell sorting and high-resolution imaging.
“Putting all of these components together into a single, reliable system was the key,” says first author Kimmo Rantalainen, a senior scientist in Schief’s lab. “The individual pieces already existed, but making them work together in a way that’s reproducible and scalable opens up new possibilities for how vaccines are analyzed and designed.”
New Insights Into Antibody Responses
Using HIV as an example, the researchers focused on a stable region of the virus’s surface protein located near the membrane. This region is targeted by a group of antibodies that can block a wide range of HIV variants. These antibodies recognize parts of the virus that remain consistent even as it mutates, making them especially valuable for vaccine research.
With the nanodisc platform, the team captured detailed structural views of how these antibodies interact with viral proteins in their natural membrane environment. This revealed features that cannot be seen when proteins are studied in isolation. The findings also shed light on how certain antibodies may neutralize viruses by disrupting the structures they use to infect cells, offering useful clues for designing better vaccines.
“The structure gave us a level of detail we simply couldn’t access before,” notes Rantalainen. “It showed us new interactions at the membrane interface and suggested why those matter for antibody function.”
Applications Beyond HIV and Ebola
To show that the method is broadly useful, the researchers also applied it to Ebola proteins. The results confirmed that antibodies could successfully recognize and bind to these proteins within the same membrane-like environment.
The platform is not limited to structural analysis. It can also be used to study immune responses to vaccine candidates. By using nanodiscs as molecular “bait,” scientists can isolate immune cells that respond to specific viral proteins. This provides a clearer understanding of how the body reacts to different vaccine designs. In addition, the system is efficient. Processes that once took a month or more can now be completed in about a week, making it easier to compare multiple vaccine candidates.
A Tool to Accelerate Vaccine Development
While the platform itself is not a vaccine, it serves as a powerful tool to support vaccine research. This is especially important for viruses that have been difficult to target using traditional methods.
“This gives the field a more realistic, accurate way to test ideas early on,” emphasizes Schief. “By improving how we study viral proteins and antibody responses, we hope this platform will help advance next-generation vaccines against some of the world’s most challenging viruses.”
In addition to Schief and Rantalainen, authors of the study “Virus glycoprotein nanodisc platform for vaccine analytics,” include Alessia Liguori, Gabriel Ozorowski, Claudia Flynn, Jon M. Steichen, Olivia M. Swanson, Patrick J. Madden, Sabyasachi Baboo, Swastik Phulera, Anant Gharpure, Danny Lu, Oleksandr Kalyuzhniy, Patrick Skog, Sierra Terada, Monolina Shil, Jolene K. Diedrich, Erik Georgeson, Ryan Tingle, Saman Eskandarzadeh, Wen-Hsin Lee, Nushin Alavi, Diana Goodwin, Michael Kubitz, Sonya Amirzehni, Devin Sok, Jeong Hyun Lee, John R. Yates III, James C. Paulson, Shane Crotty, Torben Schiffner and Andrew B. Ward of Scripps Research; and Sunny Himansu of Moderna Inc.
This work was supported by funding from the National Institute of Allergy and Infectious Diseases of the National Institutes of Health (grants UM1 AI144462, R01 AI147826, R56 AI192143 and 5F31AI179426-02); the Bill and Melinda Gates Foundation Collaboration for AIDS Vaccine Discovery (grants INV-007522, INV-008813 and INV-002916); the IAVI Neutralizing Antibody Center (INV-034657 and INV-064772); and the Alexander von Humboldt Foundation.